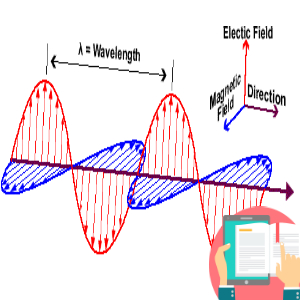
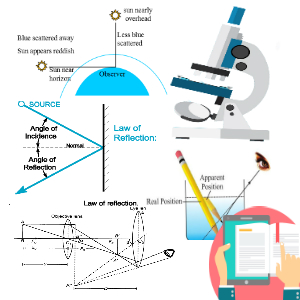
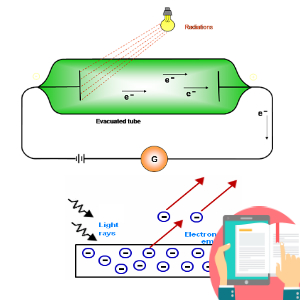
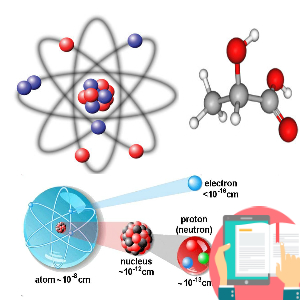
1. Question set 1 2. Question set 2 3. Question set 3 4. Question set 4 5. Question set 5 6. Question set 6 7. Question set 7 8. Question set 8 9. Question set 9 10. Question set 10 11. Question set 11 12. Question set 12 13. Question set 13
II – Answer the following1. Question set 1 2. Question set 2 3. Question set 3 4. Question set 4 5. Question set 5 6. Question set 6 7. Question set 7 8. Question set 8 9. Question set 9 10. Question set 10 11. Question set 11 12. Question set 12 13. Question set 13 14. Question set 14 15. Question set 15 16. Question set 16 17. Question set 17 18. Question set 18 19. Question set 19 20. Question set 20 21. Question set 21 22. Question set 22 23. Question set 23 24. Question set 24 25. Question set 25 26. Question set 26 27. Question set 27
1. Question set 1 2. Question set 2 3. Question set 3 4. Question set 4 5. Question set 5 6. Question set 6 7. Question set 7 8. Question set 8 9. Question set 9 10. Question set 10 11. Question set 11 12. Question set 12 13. Question set 13 14. Question set 14 15. Question set 15 16. Question set 16 17. Question set 17 18. Question set 18 19. Question set 19 20. Question set 20 21. Question set 21 22. Question set 22 23. Question set 23 24. Question set 24 25. Question set 25 26. Question set 26 27. Question set 27 28. Question set 28 29. Question set 29 30. Question set 30 31. Question set 31 32. Question set 32 33. Question set 33 34. Question set 34 35. Question set 35 36. Question set 36 37. Question set 37
1. Question set 1 2. Question set 2 3. Question set 3 4. Question set 4 5. Question set 5 6. Question set 6 7. Question set 7
1. Question set 1 2. Question set 2 3. Question set 3 4. Question set 4 5. Question set 5 6. Question set 6 7. Question set 7 8. Question set 8 9. Question set 9 10. Question set 10 11. Question set 11
II – Answer the following1. Question set 1 2. Question set 2 3. Question set 3 4. Question set 4 5. Question set 5 6. Question set 6 7. Question set 7 8. Question set 8 9. Question set 9 10. Question set 10
1. Question set 1 2. Question set 2 3. Question set 3 4. Question set 4 5. Question set 5 6. Question set 6 7. Question set 7 8. Question set 8 9. Question set 9 10. Question set 10 11. Question set 11 12. Question set 12 13. Question set 13 14. Question set 14 15. Question set 15 16. Question set 16 17. Question set 17 18. Question set 18 19. Question set 19 20. Question set 20 21. Question set 21 22. Question set 22
1. Write a short notes on the prisms making use of total internal reflection. 2. What is Snell’s window? 3. Write a note on optical fibre. 4. Explain the working of an endoscope. 5. What are primary focus and secondary focus of concex lens? 6. What are the sign conventions followed for lenses? 7. Arrive at lens equation from lens maker’s formula. 8. Obtain the equation for lateral magnification for thin lens. 9. What is power of a lens? 10. Derive the equation for effective focal length for lenses in contact. 11. What is angle of minimum deviation? 12. What is dispersion? 13. How are rainbows formed? 14. What is Rayleigh’s scattering? 15. Why does sky appear blue? 16. What is the reason for reddish appearance of sky during sunset and sunrise? 17. Why do clouds appear white? 18. What are the salient features of corpuscular theory of light? 19. What is wave theory of light? 20. What is electromagnetic wave theory of light? 21. Write a short note on quantum theory of light.
Long Answer Questions1. Derive the equation for lateral displacement of light passing through a glass slab. 2. Derive equation for refraction at single spherical surface. 3. Obtain an equation for lateral magnification due to single spherical surface. 4. Obtain Lens maker formula and mention its significance. 5. Derive the equation for angle of deviation produced by prism and thus obtain the equation for refractive index of material of the prism. 6. Derive the equation for effective focal length for lens in contact. 7. Derive the equation for effective focal length for lenses in out of contact. 8. Derive the equation for angle of deviation produced by prism and thus obtain the equation for refractive index of material of the prism. 9. What is dispersion? Obtain the equation for dispersive power of medium. 10. Prove laws of reflection using Huygens principle. 11. Prove laws of refraction using Huygen’s principle. 12. obtain the equation for resultant intensity due to interference of light. 13. Obtain the equation of path difference and band width in Young’s double slit experiment. 14. Obtain the equation for constructive and destructive interference for transmitted and reflected waves in thin films. 15. Discuss the diffraction at single slit and obtain the condition of nth minimum. 16. Discuss the diffraction at a grating and obtain the condition for nth maximum. 17. Discuss the experiment to determine the wavelength of monochromatic light using the diffraction grating. 18. Discuss the experiment to determine the wavelength of different colours using diffraction grating. 19. Explain about compound microscope and obtain the equation for magnification. 20. Discuss about the astronomical telescope. 21. Explain the experimental determination of material of the prism using spectrometer.
Conceptual Questions1. Why are dish antennas curved? 2. What type of lens is formed by a bubble inside water? 3. It is possible for two lenses to produce zero power? 4. Why does sky look blue and clouds look white? 5. Why is yellow light preferred to during fog? 6. Two independent monochromatic sources cannot act as coherent sources, why? 7. Does diffraction take place at the Young’s double slit? 8. Is there any difference between colored light obtained from prism and colours of soap bubble? 9. A small disc is placed in the path of the light from distance source. Will the center of the shadow be bright or dark? 10. When a wave undergoes reflection at a denser medium, what happens to its phase?
1. Question set 1 2. Question set 2 3. Question set 3 4. Question set 4 5. Question set 5 6. Question set 6 7. Question set 7 8. Question set 8 9. Question set 9 10. Question set 10 11. Question set 11 12. Question set 12 13. Question set 13
Very short answer1. Question set 1 2. Question set 2 3. Question set 3 4. Question set 4 5. Question set 5 6. Question set 6 7. Question set 7 8. Question set 8 9. Question set 9
Short answer1. Question set 1 2. Question set 2 3. Question set 3 4. Question set 4 5. Question set 5 6. Question set 6
1.What are cathode rays? 2.Write the properties of cathode rays. 3.Give the results of Rutherford alpha scattering experiment. 4.Write down the postulates of Bohr atom model. 5.What is meant by excitation energy? 6.Define the ionization energy and ionization potential. 7.Write down the draw backs of Bohr atom model. 8.What is distance of closest apporach? 9.Define impact parameter. 10.Write a general notation of nucleus of element X.What each term denotes? 11.What is isotope?Give an example. 12.What is isotone?Give an example. 13.What is isobar?Give an example. 14.Define atomic mass unit u. 15.Show that nuclear density is almost constant for nuclei with Z>10. 16.What is mass defect? 17.What is binding energy of a nucleus?Give its expression. 18.Calculate the energy equivalent of 1 atomic mass unit. 19.Give the physical meaning of binding energy per nucleon. 20.What is meant by radioactivity? 21.Give the symbolic representation of alpha decay,beta decay and gamma decay. 22.In alpha decay,why the unstable nucleus emits24 He nucleus?Why it does not emit four separate nucleons? 23.What is mean life of nucleus?Give the expression. 24.What is half-life of nucleus?Give the expression. 25.What is meant by activity or decay rate?Give its unit. 26.Define curie 27.What are they constituent particles of neutron and proton?
Long Answer Questions1. Explain the J.J. Thomson experiment to determine the specific charge of electron. 2. Discuss the Millikan’s oil drop experiment to determine the charge of an electron. 3. Derive the energy expression for hydrogen atom using Bohr atom model. 4. Discuss the spectral series of hydrogen atom. 5. Explain the variation of average binding energy with the mass number by graph and discuss its features. 6. Explain in detail the nuclear force. 7. Discuss the alpha decay process with example. 8. Discuss the beta decay process with examples. 9. Discuss the gamma decay process with example. 10. Obtain the law of radioactivity. 11. Discuss the properties of neutrino and its role in beta decay. 12. Explain the idea of carbon dating. 13. Discuss the process of nuclear fission and its properties. 14. Discuss the process of nuclear fusion and how energy is generated in stars? 15. Describe the working of nuclear reactor with a block diagram. 16. Explain in detail the four fundamental forces. 17. Briefly explain the elementary particles of nature.
Exercises
1. Consider two hydrogen atoms HA and HB in ground state. Assume that hydrogen atom HA is at rest and hydrogen atom HB is moving with a speed and make head-on collide on the stationary hydrogen atom HA. After the strike, both of them move together. What is minimum value of the kinetic energy of the moving hydrogen atom HB, such that any one of the hydrogen atoms reaches one of the excitation state.
2. In the Bohr atom model, the frequency of transitions is given by the following expression v=Rc((1/n2)-(1/m2)), where n<m, Consider the following transitions:
Show that the frequency of these transitions obey sum rule (which is known as Ritz combination principle)
3. (a) A hydrogen atom is excited by radiation of wavelength 97.5 nm. Find the principal quantum number of the excited state.
(b) Show that the total number of lines in emission spectrum is (n(n-1))/2 and compute the total number of possible lines in emission spectrum.
4. Calculate the radius of the earth if the density of the earth is equal to the density of the nucleus.[mass of earth 5.97 × 1024 kg ].
5. Calculate the mass defect and the binding energy per nucleon of the 10847Ag nucleus. [atomic mass of Ag = 107.905949]
6. Half lives of two radioactive elements A and B are 20 minutes and 40 minutes respectively. Initially, the samples have equal number of nuclei. Calculate the ratio of decayed numbers of A and B nuclei after 80 minutes.
7. On your birthday, you measure the activity of the sample 210Bi which has a half-life of 5.01 days. The initial activity that you measure is 1µCi . (a) What is the approximate activity of the sample on your next birthday? Calculate (b) the decay constant (c) the mean life (d) initial number of atoms.
8. Calculate the time required for 60% of a sample of radon undergo decay. Given T1/2 of radon =3.8 days
9. Assuming that energy released by the fission of a single 92235U nucleus is 200MeV, calculate the number of fissions per second required to produce 1 watt power.
10. Show that the mass of radium ( 88226Ra ) with an activity of 1 curie is almost a gram. Given T1/2 =1600 years.
11. Charcoal pieces of tree is found from an archeological site. The carbon-14 content of this charcoal is only 17.5% that of equivalent sample of carbon from a living tree. What is the age of tree?
1. Question set 1 2. Question set 2 3. Question set 3 4. Question set 4 5. Question set 5 6. Question set 6 7. Question set 7 8. Question set 8
1. Question set 1 2. Question set 2 3. Question set 3 4. Question set 4 5. Question set 5 6. Question set 6 7. Question set 7 8. Question set 8 9. Question set 9 10. Question set 10 11. Question set 11 12. Question set 12 13. Question set 13 14. Question set 14 15. Question set 15
Long Answer1. Question set 1 2. Question set 2 3. Question set 3 4. Question set 4 5. Question set 5 6. Question set 6 7. Question set 7 8. Question set 8 9. Question set 9 10. Question set 10
Extra Questions1. Question set 1 2. Question set 2 3. Question set 3
1. Question set 1 2. Question set 2 3. Question set 3 4. Question set 4 5. Question set 5 6. Question set 6 7. Question set 7
Long Answer1. Question set 1 2. Question set 2 3. Question set 3 4. Question set 4 5. Question set 5
Extra Questions1. Question set 1 2. Question set 2 3. Question set 3 4. Question set 4 5. Question set 5 6. Question set 6